Time:2025-08-26

Recently, I saw an article titled "Is moisture determination that simple?" on the Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng
Sulindac (Clindamycin) 2% Ointment is a novel small-molecule, non-hormonal, non-steroidal anti-inflammatory agent that inhibits phosphodiesterase-4 (PDE-4). PDE-4 mediates the conversion of cyclic adenosine monophosphate (cAMP) to monophosphoadenosine (AMP), thereby reducing intracellular cAMP levels. In physiology, elevated cAMP concentrations in T cells and other immune cells inhibit the production of inflammatory mediators. Patients with atopic dermatitis exhibit low cAMP levels and abnormally high PDE activity in white blood cells and monocytes, with increased PDE activity associated with inflammatory responsiveness. Inhibiting PDE reduces the release of pro-inflammatory cytokines.
Figure 1: Structure of Kliborol
I. Differences in KF and LOD data for Clebromol
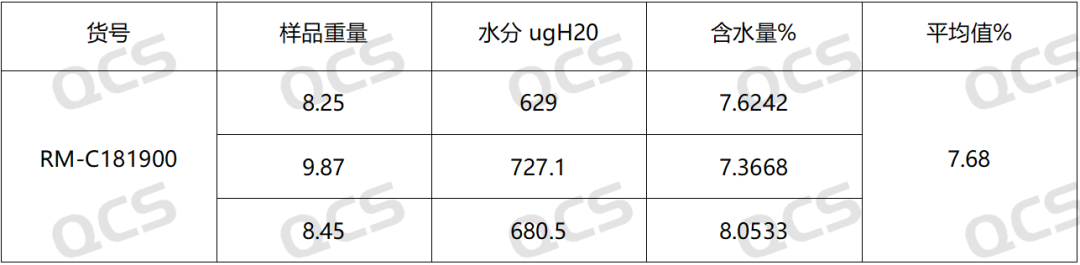
Table 1.KF data

Table 2.LOD data
According to the data of the two methods, the results of moisture detection are 7% different and the data do not correspond.
II. CAUSE ANALYSIS
Boron-containing compounds, especially organic borates and borates (borates) (cliborro belongs to this category), are often encountered in significant interference in KF determination, resulting in high results and a large difference from the LOD results:
Lewis acidity of boron atoms
Boron atoms (especially trivalent boron) have empty p orbitals and are strong Lewis acids.
The key components of the Karl Fischer reagent (ROH-methanol/ethanol, Base-pyridine/imidazole) are all Lewis bases.
Main interference mechanism:
Formation of Complexes: Boron atoms coordinate with alkali (e.g., imidazole) or alcohol (RO⁻) in the KF reagent to form stable complexes. While these reactions themselves do not consume water, they disrupt the chemical equilibrium of the reaction system or deplete active components in the KF reagent. This results in difficulties in determining the titration endpoint or requiring additional iodine to reach the endpoint, ultimately leading to overestimation of the measured moisture content.
Hydrolysis/Alcoholysis reactions: Certain borate esters (e.g., the borate bonds in cliborol) may become unstable in a KF-containing environment (which may contain alcohol and trace amounts of water), leading to hydrolysis or alcoholysis. These reactions could release additional water molecules (from either the sample itself or the environment) or generate substances that react with iodine, which would similarly result in artificially elevated test values.
Depletion of iodine by the secondary reaction: In addition to the main reaction, boron compounds or their decomposition products may react with iodine in other REDOX reactions and consume iodine non-specifically, resulting in the instrument mistakenly thinking that the iodine consumed in this part is also used for titration of water, thus resulting in a higher measured moisture value than the true value.
The reason for the difference between KF and LOD:
The KF method results are high: the above interference mechanism (mainly complexation reaction and non-water side reaction consumption of iodine) leads to the "water" value measured by KF method is significantly higher than the actual physical bound water content in the sample.
LOD Method Results: The Loss on Dry Matter (LOD) method measures the total mass loss of a sample after heating at a specific temperature and duration. This loss includes not only moisture but also volatile solvents, low-boiling-point components, and even volatile byproducts generated during thermal decomposition. For complex molecules like cloroborophane, the likelihood of thermal decomposition is significantly higher.
Root causes of variance:
KF method: the "water" result is exaggerated due to chemical interference (including false water signal caused by side reactions).
LOD method: it reflects the total loss of volatile substances, which may underestimate the true moisture (if some water is tightly bound and not easy to dry out) or overestimate the moisture (if it contains other volatile substances or decomposition products).
conclusion
For boron-containing compounds such as clibor, the measured value of KF method is usually much higher than that of LOD method due to severe chemical interference, and neither of them can accurately represent the real physical bound water content.
The high result of KF was caused by interference, while the LOD result was affected by thermal stability and volatile composition. The difference was mainly due to the specific interference of KF method.
III. Other types of samples that cannot be tested for moisture with Karl Fischer reagent
In addition to boron-containing compounds, there are many types of samples that interfere with KF determination, resulting in inaccurate or invalid results:
Strong reducing substance
Examples: thiosulfinates (R-SH), sulfides (H₂S, R-S-R'), sulfites, tin(II) salts, ascorbic acid, etc.
Problem: These substances can directly react with iodine (I₂) in KF reagent, consume iodine, and lead to an overestimation of the water value.
Strong oxidizing substances
Examples: peroxide (H₂O₂, ROOR), high-valent metal oxides (e.g. CrO₃, MnO₂), iodides, quinones, etc.
Problem: These substances can oxidize iodide (I⁻) in KF reagent to iodine (I₂), resulting in the generation of additional iodine before or during the titration, resulting in an early or unattainable endpoint of titration, resulting in low water value or unstable results.
Compounds that can react with components of the KF reagent
Aldehydes and ketones: Particularly low-grade aldehydes (such as formaldehyde) are prone to undergo condensation aldehyde reaction with methanol in KF reagent, releasing water molecules (RCHO + 2CH₃OH → RCH(OCH₃)₂ + H₂O), resulting in an overestimated moisture content. Ketones may also exhibit similar but weaker reactions.
L活泼硅烷/siloxane: may release hydrogen or react with reagents.
Metal hydroxide/oxide: such as quicklime (CaO), caustic alkali (NaOH, KOH), which react with water or absorb water/CO₂ themselves, are difficult to treat and may interfere with the reaction.
A compound that produces water
Examples: carboxylic anhydride (RCO) ₂O + H ₂O → 2RCOOH), reaction of strong acid with alcohol (esterification), certain condensation reactions.
Problem: These compounds slowly react with water or with each other in the KF reagent environment to form water, resulting in a time-dependent increase in moisture value and unreliable results.
Insoluble sample or reactive solid
Examples: some metals, metal oxides, carbonates, desiccants, etc.
Problem: Water may be wrapped up and difficult to release completely; the solid surface may have a side reaction with the reagent (e.g., carbonate reacts with acid to produce CO₂, which may interfere with the endpoint detection).
Compounds containing active metals
Examples: organometallic compounds (Grignard reagents, alkyl lithium, etc.).
Problem: extremely active, may react violently with any proton source in the reagent (including trace water, alcohol), even dangerous, and interfere seriously.
IV. CONCLUSION
As a boron-containing organic compound, the boron center with Lewis acidity of cliborol will complex with the Lewis base component (alcohol and alkali) in Karl Fischer reagent or induce side reactions, which will non-specifically consume iodine or change the reaction balance, resulting in significantly high water value measured by KF method.
The LOD method measures the total volatile loss, which may include water, solvent and decomposition products. The two methods have different principles and different interfering factors, so the results are different.
Although KF method is the standard for water determination, it has inherent limitations for many types of samples such as boron-containing compounds, strong oxidizing/reducing agents, aldehydes and ketones, and substances that can generate water. Therefore, sample compatibility must be considered when selecting the method.



Recently, I saw an article titled "Is moisture determination that simple?" on the Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng Weizheng
Sulindac (Clindamycin) 2% Ointment is a novel small-molecule, non-hormonal, non-steroidal anti-inflammatory agent that inhibits phosphodiesterase-4 (PDE-4). PDE-4 mediates the conversion of cyclic adenosine monophosphate (cAMP) to monophosphoadenosine (AMP), thereby reducing intracellular cAMP levels. In physiology, elevated cAMP concentrations in T cells and other immune cells inhibit the production of inflammatory mediators. Patients with atopic dermatitis exhibit low cAMP levels and abnormally high PDE activity in white blood cells and monocytes, with increased PDE activity associated with inflammatory responsiveness. Inhibiting PDE reduces the release of pro-inflammatory cytokines.

Figure 1: Structure of Kliborol
I. Differences in KF and LOD data for Clebromol

Table 1.KF data

Table 2.LOD data
According to the data of the two methods, the results of moisture detection are 7% different and the data do not correspond.
II. CAUSE ANALYSIS
Boron-containing compounds, especially organic borates and borates (borates) (cliborro belongs to this category), are often encountered in significant interference in KF determination, resulting in high results and a large difference from the LOD results:
Lewis acidity of boron atoms
Boron atoms (especially trivalent boron) have empty p orbitals and are strong Lewis acids.
The key components of the Karl Fischer reagent (ROH-methanol/ethanol, Base-pyridine/imidazole) are all Lewis bases.
Main interference mechanism:
Formation of Complexes: Boron atoms coordinate with alkali (e.g., imidazole) or alcohol (RO⁻) in the KF reagent to form stable complexes. While these reactions themselves do not consume water, they disrupt the chemical equilibrium of the reaction system or deplete active components in the KF reagent. This results in difficulties in determining the titration endpoint or requiring additional iodine to reach the endpoint, ultimately leading to overestimation of the measured moisture content.
Hydrolysis/Alcoholysis reactions: Certain borate esters (e.g., the borate bonds in cliborol) may become unstable in a KF-containing environment (which may contain alcohol and trace amounts of water), leading to hydrolysis or alcoholysis. These reactions could release additional water molecules (from either the sample itself or the environment) or generate substances that react with iodine, which would similarly result in artificially elevated test values.
Depletion of iodine by the secondary reaction: In addition to the main reaction, boron compounds or their decomposition products may react with iodine in other REDOX reactions and consume iodine non-specifically, resulting in the instrument mistakenly thinking that the iodine consumed in this part is also used for titration of water, thus resulting in a higher measured moisture value than the true value.
The reason for the difference between KF and LOD:
The KF method results are high: the above interference mechanism (mainly complexation reaction and non-water side reaction consumption of iodine) leads to the "water" value measured by KF method is significantly higher than the actual physical bound water content in the sample.
LOD Method Results: The Loss on Dry Matter (LOD) method measures the total mass loss of a sample after heating at a specific temperature and duration. This loss includes not only moisture but also volatile solvents, low-boiling-point components, and even volatile byproducts generated during thermal decomposition. For complex molecules like cloroborophane, the likelihood of thermal decomposition is significantly higher.
Root causes of variance:
KF method: the "water" result is exaggerated due to chemical interference (including false water signal caused by side reactions).
LOD method: it reflects the total loss of volatile substances, which may underestimate the true moisture (if some water is tightly bound and not easy to dry out) or overestimate the moisture (if it contains other volatile substances or decomposition products).
conclusion
For boron-containing compounds such as clibor, the measured value of KF method is usually much higher than that of LOD method due to severe chemical interference, and neither of them can accurately represent the real physical bound water content.
The high result of KF was caused by interference, while the LOD result was affected by thermal stability and volatile composition. The difference was mainly due to the specific interference of KF method.
III. Other types of samples that cannot be tested for moisture with Karl Fischer reagent
In addition to boron-containing compounds, there are many types of samples that interfere with KF determination, resulting in inaccurate or invalid results:
Strong reducing substance
Examples: thiosulfinates (R-SH), sulfides (H₂S, R-S-R'), sulfites, tin(II) salts, ascorbic acid, etc.
Problem: These substances can directly react with iodine (I₂) in KF reagent, consume iodine, and lead to an overestimation of the water value.
Strong oxidizing substances
Examples: peroxide (H₂O₂, ROOR), high-valent metal oxides (e.g. CrO₃, MnO₂), iodides, quinones, etc.
Problem: These substances can oxidize iodide (I⁻) in KF reagent to iodine (I₂), resulting in the generation of additional iodine before or during the titration, resulting in an early or unattainable endpoint of titration, resulting in low water value or unstable results.
Compounds that can react with components of the KF reagent
Aldehydes and ketones: Particularly low-grade aldehydes (such as formaldehyde) are prone to undergo condensation aldehyde reaction with methanol in KF reagent, releasing water molecules (RCHO + 2CH₃OH → RCH(OCH₃)₂ + H₂O), resulting in an overestimated moisture content. Ketones may also exhibit similar but weaker reactions.
L活泼硅烷/siloxane: may release hydrogen or react with reagents.
Metal hydroxide/oxide: such as quicklime (CaO), caustic alkali (NaOH, KOH), which react with water or absorb water/CO₂ themselves, are difficult to treat and may interfere with the reaction.
A compound that produces water
Examples: carboxylic anhydride (RCO) ₂O + H ₂O → 2RCOOH), reaction of strong acid with alcohol (esterification), certain condensation reactions.
Problem: These compounds slowly react with water or with each other in the KF reagent environment to form water, resulting in a time-dependent increase in moisture value and unreliable results.
Insoluble sample or reactive solid
Examples: some metals, metal oxides, carbonates, desiccants, etc.
Problem: Water may be wrapped up and difficult to release completely; the solid surface may have a side reaction with the reagent (e.g., carbonate reacts with acid to produce CO₂, which may interfere with the endpoint detection).
Compounds containing active metals
Examples: organometallic compounds (Grignard reagents, alkyl lithium, etc.).
Problem: extremely active, may react violently with any proton source in the reagent (including trace water, alcohol), even dangerous, and interfere seriously.
IV. CONCLUSION
As a boron-containing organic compound, the boron center with Lewis acidity of cliborol will complex with the Lewis base component (alcohol and alkali) in Karl Fischer reagent or induce side reactions, which will non-specifically consume iodine or change the reaction balance, resulting in significantly high water value measured by KF method.
The LOD method measures the total volatile loss, which may include water, solvent and decomposition products. The two methods have different principles and different interfering factors, so the results are different.
Although KF method is the standard for water determination, it has inherent limitations for many types of samples such as boron-containing compounds, strong oxidizing/reducing agents, aldehydes and ketones, and substances that can generate water. Therefore, sample compatibility must be considered when selecting the method.


Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*All our products are for R&D.

*All our products are for R&D.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号