Time:2026-04-15


In March 2026, Appco Pharma LLC voluntarily recalled 175,061 bottles of prazosin hydrochloride capsules (specifications: 1 mg, 2 mg, 5 mg). Just five months prior, in October 2025, Teva Pharmaceuticals had also recalled over 580,000 bottles of the same drug.
The question arises: What prompted both companies to independently initiate safety alerts following separate product recalls from different manufacturers? In fact, this was no coincidence—the true culprit was the excessive presence of N-nitroso-prazosin EP Impurity C.The question arises: What prompted both companies to independently initiate safety alerts following separate product recalls from different manufacturers? In fact, this was no coincidence—the true culprit was the excessive presence of N-nitroso-prazosin EP Impurity C.

01 N-nitroso-piperazosine EP Impurity C: Chemical Identity and Structural Relationship
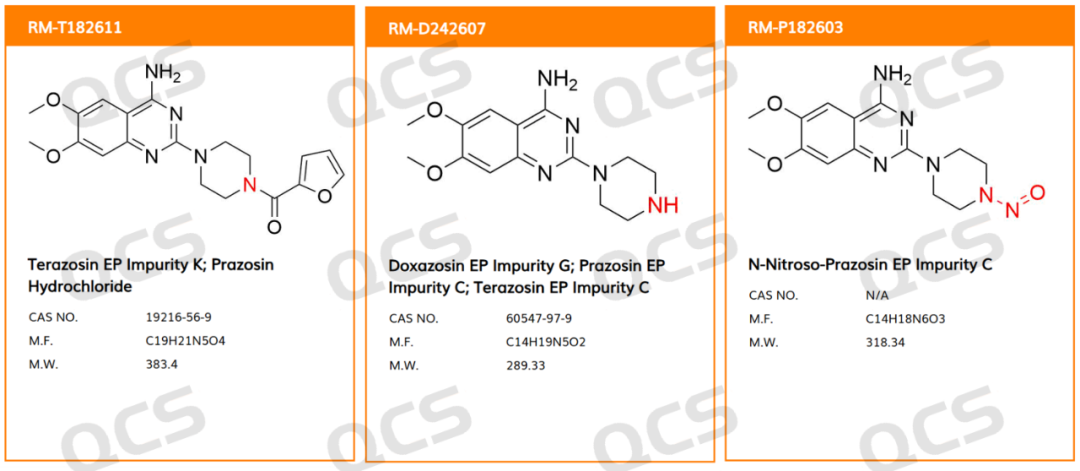
The primary cause of this recall is N-nitroso-prazosin EP Impurity C, which is a nitrosamine derivative of Prazosin EP Impurity C.
Prazosin EP Impurity C: Non-nitroso form; QCS Product Code: RM-D242607Prazosin EP Impurity C: Non-nitroso form; QCS Product Code: RM-D242607
6,7-Dimethoxy-2-(piperazin-1-yl)quinazolin-4-amine is also an impurity of doxazosin (EP Impurity G) and terazosin (EP Impurity C). Since EP Impurity C of哌唑嗪 contains a哌嗪 (piperazine) ring structure, it is essentially a secondary amine. Compared to the active pharmaceutical ingredient, it is more susceptible to nitrosamination reactions and serves as a potential nitrosamine precursor.
The source of the precursor prazosin impurity C is extensive, potentially including residual intermediates, degradation of the active pharmaceutical ingredient (API), or secondary formation in the formulation environment. The presence of prazosin EP impurity C is often difficult to avoid.
N-nitroso-piperaz EP impurity C: nitroso form; QCS product code: RM-P182603
These are the N-nitrosyl derivatives of the aforementioned non-nitrosyl impurities (where the nitrogen atom of the piperazine ring undergoes N-nitrosylation). Since the EP impurity C of唑嗪 is also present in both doxazosin and terazosin, the N-nitrosyl-piperazosin EP impurity C may also exist in these drugs. Therefore, this impurity requires attention in the research of these medications.
02 Formation Mechanism and Risk Characteristics of Specific Nitrosamines
3.1 Nitrosation Reaction Path
During API synthesis or formulation storage, prazosin or its non-nitroso impurities (containing secondary/tertiary amine fragments) may react with nitrosating reagents (such as nitrites, which may originate from excipients, packaging materials, or the environment):
R2NH+NO2-(Acidic conditions)→R2N–NO
This reaction occurs most readily under acidic pH conditions (particularly pH 3–4). For the piperazine ring, nitrosation can occur on any nitrogen atom, yielding N-nitroso-piperazine derivatives.
3.2 Special Risks of NDSRI
· Dual origin of impurities: These may result either from process-related impurities introduced during API synthesis or from secondary degradation impurities formed in the formulation.Dual origin of impurities: These may result either from process-related impurities introduced during API synthesis or from secondary degradation impurities formed in the formulation.
· Shelf-life growth: Some NDSRIs exhibited an upward trend during finished product stability studies, indicating the need to monitor the formulation environment (moisture, pH, excipients, and temperature).
· Structural activity uncertainty: Most NDSRs lack comprehensive toxicological data. Regulatory agencies employ the CPCA (Carcinogenic Potency Categorization Approach) to predict carcinogenic potency based on structural similarity and determine provisional limits (e.g., the 5 ppm limit adopted during the prazosin recall was derived from CPCA).
03 Regulatory Requirements

The two consecutive recalls of prazosin clearly highlight the complexity and urgency of NDSRI control—impurities may originate from the API or may 'develop' within the formulation; without reference standards, it is impossible to establish methodologies, let alone conduct reliable risk assessments.
According to the California State Board of Pharmacy, the acceptable limit for this impurity is 5 ppm. Both the 2025 and 2026 recalls were initiated by excessive levels of the N-nitroso-piperazoline EP impurity C exceeding the safety limit, leading the FDA to classify them as Class II risk recalls. The frequent recurrence of such recalls indicates that the same API, structural type, or impurity category is recurring. Of particular note: the impurities involved—N-nitroso-piperazoline EP impurity C—are not original design impurities but rather "derivative impurities."
An increasing number of examples demonstrate that regulatory agencies' focus on nitrosamine warning structures has expanded from merely assessing nitrosamines in the active pharmaceutical ingredient (API) itself to evaluating the nitrosamine risks associated with impurity components. The FDA and USP nitrosamine community emphasize that impurities may not only persist in the API but also form or accumulate in the final drug product. Consequently, for drugs containing piperazine/piperidine structures, the full-process control of nitrosamine-derived substances (NDSRIs) has become the new regulatory norm. As regulators shift their attention from NDMA to NDSRIs, high-purity, structurally well-characterized nitrosamine impurity reference standards have emerged as a critical component of the quality control framework throughout the entire lifecycle of amine-containing pharmaceuticals.
For technical specifications and quotations regarding N-nitroso-piperaz EP impurity C (RM-P182603) or other NDSRI reference standards, please contact the QCS Drug R&D Center.
QCS has obtained ANAB ISO 17034 certification, with its standard substance production and calibration processes meeting the international requirements for reference material manufacturer accreditation. This ensures data support with international mutual recognition validity for regulatory submissions to agencies such as the NMPA, EMA, and FDA.





In March 2026, Appco Pharma LLC voluntarily recalled 175,061 bottles of prazosin hydrochloride capsules (specifications: 1 mg, 2 mg, 5 mg). Just five months prior, in October 2025, Teva Pharmaceuticals had also recalled over 580,000 bottles of the same drug.
The question arises: What prompted both companies to independently initiate safety alerts following separate product recalls from different manufacturers? In fact, this was no coincidence—the true culprit was the excessive presence of N-nitroso-prazosin EP Impurity C.The question arises: What prompted both companies to independently initiate safety alerts following separate product recalls from different manufacturers? In fact, this was no coincidence—the true culprit was the excessive presence of N-nitroso-prazosin EP Impurity C.

01 N-nitroso-piperazosine EP Impurity C: Chemical Identity and Structural Relationship

The primary cause of this recall is N-nitroso-prazosin EP Impurity C, which is a nitrosamine derivative of Prazosin EP Impurity C.
Prazosin EP Impurity C: Non-nitroso form; QCS Product Code: RM-D242607Prazosin EP Impurity C: Non-nitroso form; QCS Product Code: RM-D242607
6,7-Dimethoxy-2-(piperazin-1-yl)quinazolin-4-amine is also an impurity of doxazosin (EP Impurity G) and terazosin (EP Impurity C). Since EP Impurity C of哌唑嗪 contains a哌嗪 (piperazine) ring structure, it is essentially a secondary amine. Compared to the active pharmaceutical ingredient, it is more susceptible to nitrosamination reactions and serves as a potential nitrosamine precursor.
The source of the precursor prazosin impurity C is extensive, potentially including residual intermediates, degradation of the active pharmaceutical ingredient (API), or secondary formation in the formulation environment. The presence of prazosin EP impurity C is often difficult to avoid.
N-nitroso-piperaz EP impurity C: nitroso form; QCS product code: RM-P182603
These are the N-nitrosyl derivatives of the aforementioned non-nitrosyl impurities (where the nitrogen atom of the piperazine ring undergoes N-nitrosylation). Since the EP impurity C of唑嗪 is also present in both doxazosin and terazosin, the N-nitrosyl-piperazosin EP impurity C may also exist in these drugs. Therefore, this impurity requires attention in the research of these medications.
02 Formation Mechanism and Risk Characteristics of Specific Nitrosamines
3.1 Nitrosation Reaction Path
During API synthesis or formulation storage, prazosin or its non-nitroso impurities (containing secondary/tertiary amine fragments) may react with nitrosating reagents (such as nitrites, which may originate from excipients, packaging materials, or the environment):
R2NH+NO2-(Acidic conditions)→R2N–NO
This reaction occurs most readily under acidic pH conditions (particularly pH 3–4). For the piperazine ring, nitrosation can occur on any nitrogen atom, yielding N-nitroso-piperazine derivatives.
3.2 Special Risks of NDSRI
· Dual origin of impurities: These may result either from process-related impurities introduced during API synthesis or from secondary degradation impurities formed in the formulation.Dual origin of impurities: These may result either from process-related impurities introduced during API synthesis or from secondary degradation impurities formed in the formulation.
· Shelf-life growth: Some NDSRIs exhibited an upward trend during finished product stability studies, indicating the need to monitor the formulation environment (moisture, pH, excipients, and temperature).
· Structural activity uncertainty: Most NDSRs lack comprehensive toxicological data. Regulatory agencies employ the CPCA (Carcinogenic Potency Categorization Approach) to predict carcinogenic potency based on structural similarity and determine provisional limits (e.g., the 5 ppm limit adopted during the prazosin recall was derived from CPCA).
03 Regulatory Requirements

The two consecutive recalls of prazosin clearly highlight the complexity and urgency of NDSRI control—impurities may originate from the API or may 'develop' within the formulation; without reference standards, it is impossible to establish methodologies, let alone conduct reliable risk assessments.
According to the California State Board of Pharmacy, the acceptable limit for this impurity is 5 ppm. Both the 2025 and 2026 recalls were initiated by excessive levels of the N-nitroso-piperazoline EP impurity C exceeding the safety limit, leading the FDA to classify them as Class II risk recalls. The frequent recurrence of such recalls indicates that the same API, structural type, or impurity category is recurring. Of particular note: the impurities involved—N-nitroso-piperazoline EP impurity C—are not original design impurities but rather "derivative impurities."
An increasing number of examples demonstrate that regulatory agencies' focus on nitrosamine warning structures has expanded from merely assessing nitrosamines in the active pharmaceutical ingredient (API) itself to evaluating the nitrosamine risks associated with impurity components. The FDA and USP nitrosamine community emphasize that impurities may not only persist in the API but also form or accumulate in the final drug product. Consequently, for drugs containing piperazine/piperidine structures, the full-process control of nitrosamine-derived substances (NDSRIs) has become the new regulatory norm. As regulators shift their attention from NDMA to NDSRIs, high-purity, structurally well-characterized nitrosamine impurity reference standards have emerged as a critical component of the quality control framework throughout the entire lifecycle of amine-containing pharmaceuticals.
For technical specifications and quotations regarding N-nitroso-piperaz EP impurity C (RM-P182603) or other NDSRI reference standards, please contact the QCS Drug R&D Center.
QCS has obtained ANAB ISO 17034 certification, with its standard substance production and calibration processes meeting the international requirements for reference material manufacturer accreditation. This ensures data support with international mutual recognition validity for regulatory submissions to agencies such as the NMPA, EMA, and FDA.



Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*All our products are for R&D.

*All our products are for R&D.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号