orders@qcsrm.com | +86 755-6685 3366 | 2851296953 | 13670046396
Tempo:2025-12-03

Today, we share the stability study of specific impurities in the hypoglycemic drug vildagliptin. Vildagliptin, with the chemical name 1-{[(3-hydroxy-L-金刚烷基)amino]acetyl}-2-cyanocyanol-(S)-tetrahydropyridinyl, is a selective and orally active specific dipeptidyl peptidase-4 (DPP-4) inhibitor. It inhibits DPP-4 activity by forming a DPP-4 complex, thereby increasing the concentration of glucagon-like peptide-1 (GLP-1), which promotes insulin production by pancreatic β-cells while reducing glucagon levels, thus lowering blood glucose without significant effects on body weight.
I. Experimental Design
In this experiment, our center conducted solution stability studies on three specific impurities of viglitol in accordance with the chromatographic conditions specified in the internal control standards of the enterprise. The sample numbers and structural formulas used are shown in Figures 1 and 2 below:
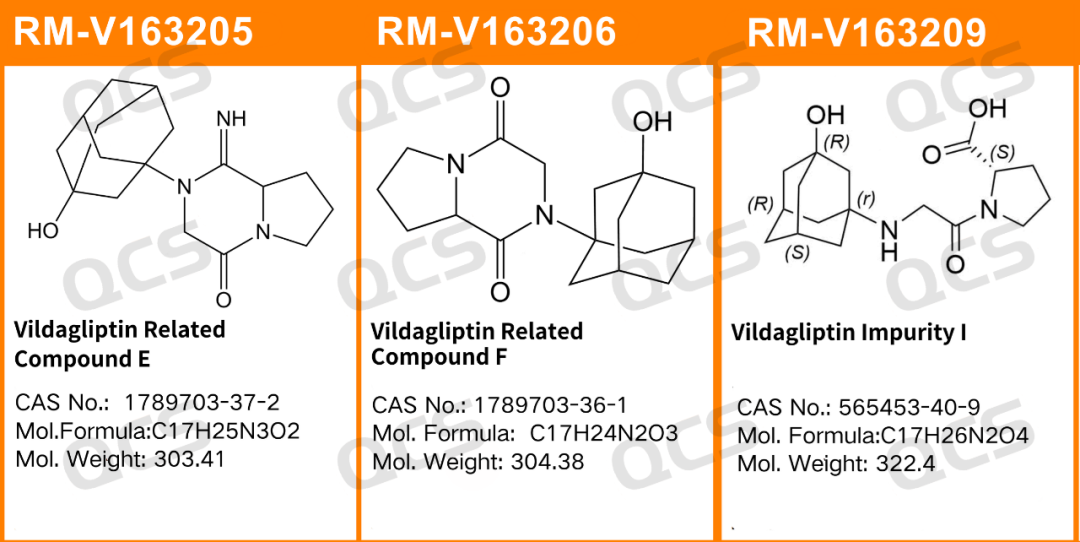
Figure 1: Impurity codes and structural formulas used in this study
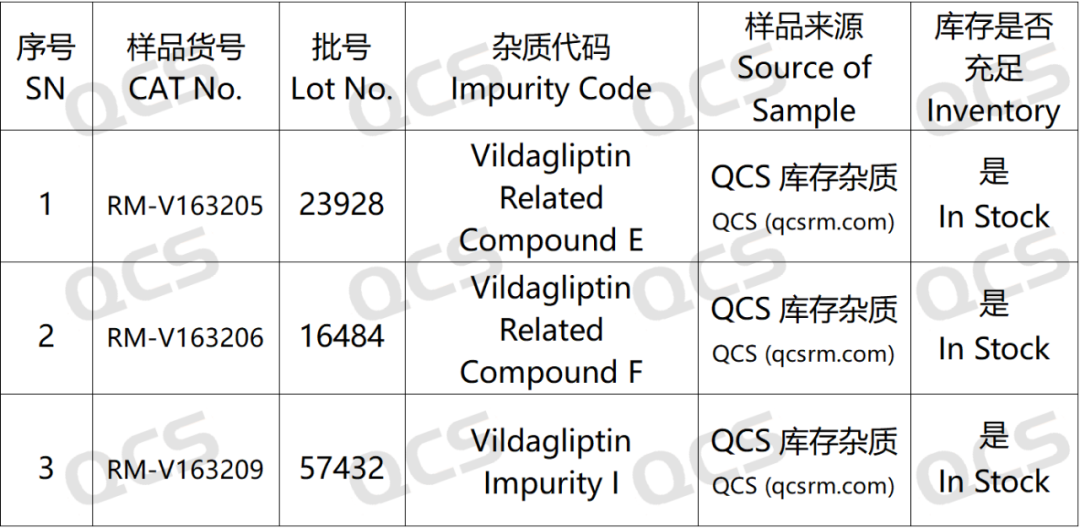
Figure 2: Correspondence between standard impurity codes and impurity product numbers used in this study
In this experiment, the laboratory technician prepared appropriate amounts of RM-V163205 (Vildagliptin Related Compound E; CAS NO: 1789703-37-2), RM-V163206 (Vildagliptin Related Compound F; CAS NO: 1789703-36-1), and RM-V163209 (Vildagliptin Impurity I; CAS NO: 565453-40-9), respectively. These samples were placed in acidic, neutral, and alkaline solutions, and stored at room temperature and pressure for 0, 3, 6, 9, 12, and 24 hours. Subsequently, the samples were analyzed by injection under the chromatographic conditions specified for the reference substance in the internal control standard of the company.The stability of the sample was determined by observing the change of the main peak area in the chromatogram with the extension of the sample solution time.
2. Experimental Conclusions
RM-V163205(Vildagliptin Related Compound E)
The test results showed that the main peak area of the sample RM-V163205 remained stable during the 24-hour storage in both acidic and neutral solutions, with relative standard deviations all below 2.0%. Therefore, it can be concluded that the sample is stable during the 24-hour storage in both acidic and neutral solutions.
However, the area of the main peak in the sample exhibited significant variation during the 24-hour exposure to alkaline solution, with a relative standard deviation of 16.32%, exceeding 2.0%. This indicates that the sample is unstable during the 24-hour alkaline solution exposure and undergoes continuous degradation over time. The area data of the main peak at each detection point under different pH conditions are as follows:
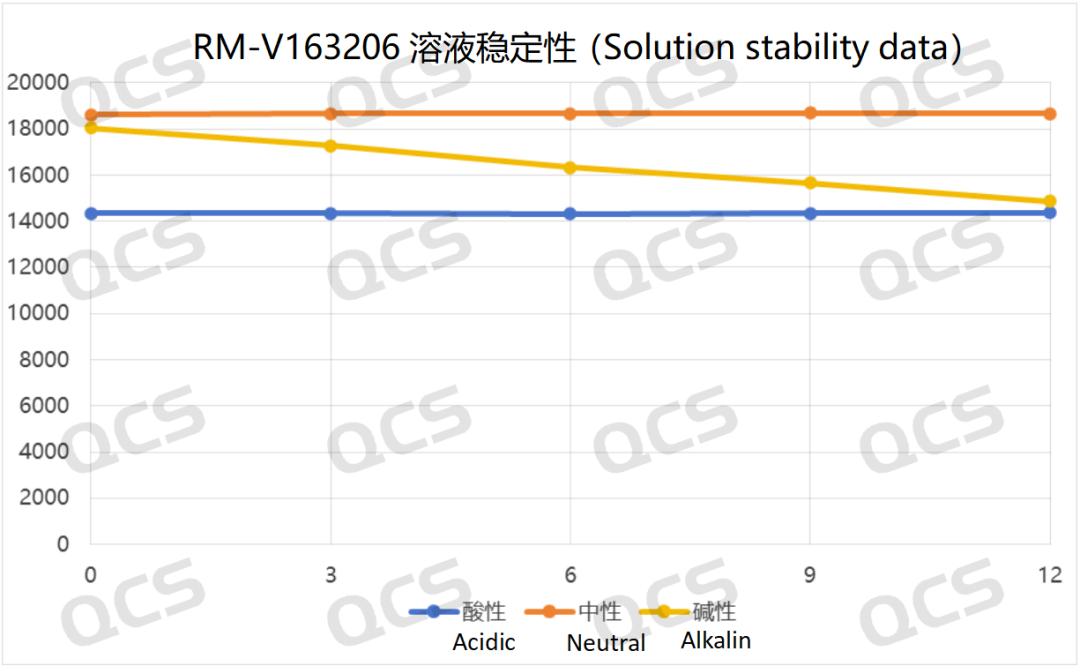
Figure 3: Summary line chart of solution stability data for sample RM-V163205 (Vildagliptin Related Compound E) after 24-hour exposure to acidic, neutral, and alkaline solutions
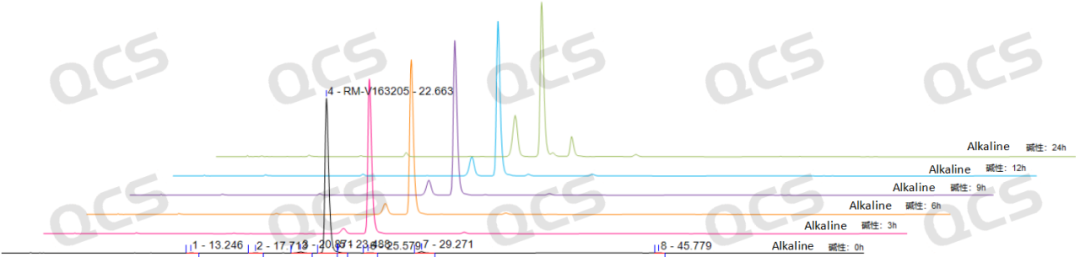
Figure 4: Stereoscopic comparison of stability data in alkaline solution for sample RM-V163205 (Vildagliptin-related compound E)
RM-V163206(Vildagliptin Related Compound F)
The test results indicate that sample RM-V163206 demonstrates minimal variation in the main peak area during 24-hour exposure to acidic, neutral, and alkaline solutions, with relative standard deviations all below 2.0%. This confirms the sample's stability across these three pH conditions. The main peak area data for each measurement point under different pH values are as follows:
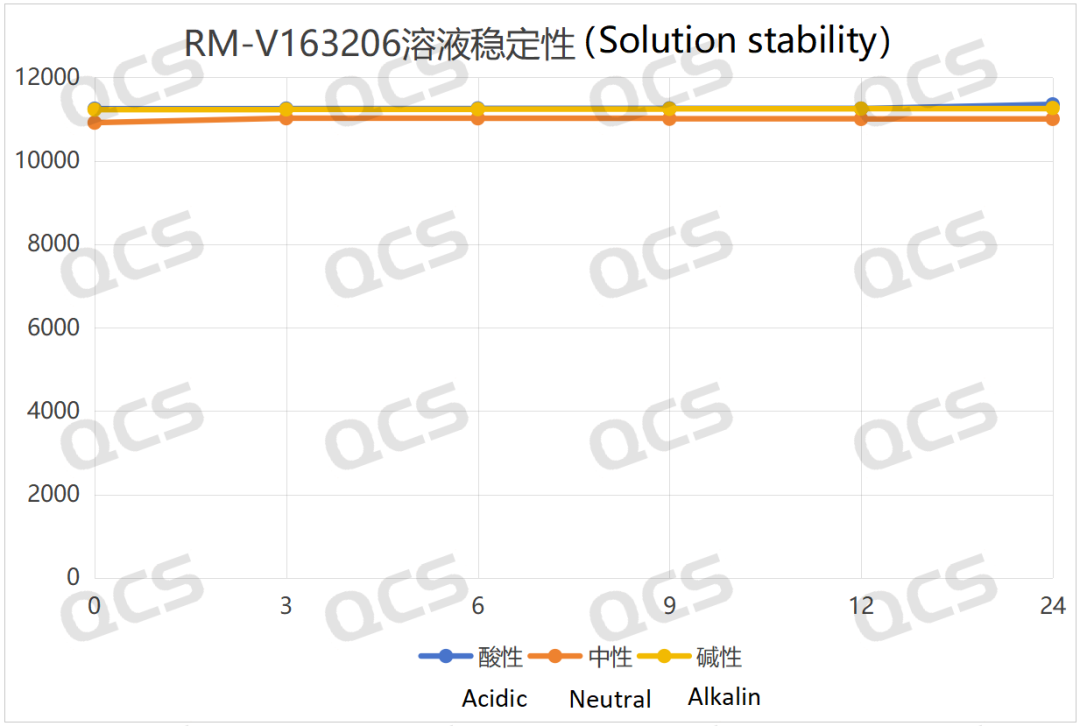
Figure 5: Summary line chart of solution stability data for sample RM-V163206 (Vildagliptin-related compound F) after 24-hour exposure to acidic, neutral, and alkaline solutions
RM-V163209(Vildagliptin Impurity I)
The test results indicate that sample RM-V163209 demonstrates minimal variation in the main peak area during 24-hour storage in acidic, neutral, and alkaline solutions, with relative standard deviations all below 2.0%. This confirms the sample's stability across these three pH conditions. The main peak area data for each measurement point under different pH values are as follows:
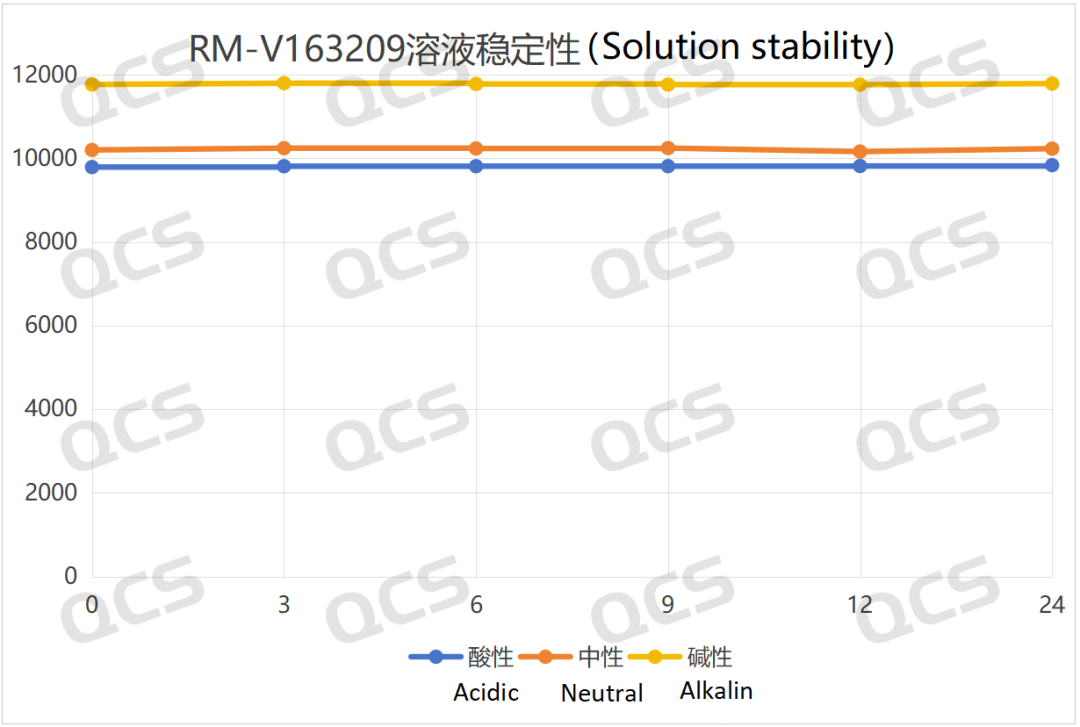
Figure 6: Summary line chart of solution stability data for sample RM-V163209 (Vildagliptin Impurity I) after 24-hour exposure to acidic, neutral, and alkaline solutions
3. Summary
In conclusion, this experiment demonstrates that samples RM-V163206 (Vildagliptin Related Compound F) and RM-V163209 (Vildagliptin Impurity I) exhibit excellent stability in acidic, neutral, and alkaline solutions. In contrast, RM-V163205 (Vildagliptin Related Compound E) shows good stability in acidic and neutral solutions but undergoes rapid degradation over time in alkaline solutions. Therefore, when testing sample RM-V163205 (Vildagliptin Related Compound E), customers should avoid using alkaline diluents and refrain from contact with alkaline reagents during handling, storage, and transportation. For further inquiries regarding the stability of these three samples, please contact our company.



Today, we share the stability study of specific impurities in the hypoglycemic drug vildagliptin. Vildagliptin, with the chemical name 1-{[(3-hydroxy-L-金刚烷基)amino]acetyl}-2-cyanocyanol-(S)-tetrahydropyridinyl, is a selective and orally active specific dipeptidyl peptidase-4 (DPP-4) inhibitor. It inhibits DPP-4 activity by forming a DPP-4 complex, thereby increasing the concentration of glucagon-like peptide-1 (GLP-1), which promotes insulin production by pancreatic β-cells while reducing glucagon levels, thus lowering blood glucose without significant effects on body weight.
I. Experimental Design
In this experiment, our center conducted solution stability studies on three specific impurities of viglitol in accordance with the chromatographic conditions specified in the internal control standards of the enterprise. The sample numbers and structural formulas used are shown in Figures 1 and 2 below:

Figure 1: Impurity codes and structural formulas used in this study

Figure 2: Correspondence between standard impurity codes and impurity product numbers used in this study
In this experiment, the laboratory technician prepared appropriate amounts of RM-V163205 (Vildagliptin Related Compound E; CAS NO: 1789703-37-2), RM-V163206 (Vildagliptin Related Compound F; CAS NO: 1789703-36-1), and RM-V163209 (Vildagliptin Impurity I; CAS NO: 565453-40-9), respectively. These samples were placed in acidic, neutral, and alkaline solutions, and stored at room temperature and pressure for 0, 3, 6, 9, 12, and 24 hours. Subsequently, the samples were analyzed by injection under the chromatographic conditions specified for the reference substance in the internal control standard of the company.The stability of the sample was determined by observing the change of the main peak area in the chromatogram with the extension of the sample solution time.
2. Experimental Conclusions
RM-V163205(Vildagliptin Related Compound E)
The test results showed that the main peak area of the sample RM-V163205 remained stable during the 24-hour storage in both acidic and neutral solutions, with relative standard deviations all below 2.0%. Therefore, it can be concluded that the sample is stable during the 24-hour storage in both acidic and neutral solutions.
However, the area of the main peak in the sample exhibited significant variation during the 24-hour exposure to alkaline solution, with a relative standard deviation of 16.32%, exceeding 2.0%. This indicates that the sample is unstable during the 24-hour alkaline solution exposure and undergoes continuous degradation over time. The area data of the main peak at each detection point under different pH conditions are as follows:

Figure 3: Summary line chart of solution stability data for sample RM-V163205 (Vildagliptin Related Compound E) after 24-hour exposure to acidic, neutral, and alkaline solutions

Figure 4: Stereoscopic comparison of stability data in alkaline solution for sample RM-V163205 (Vildagliptin-related compound E)
RM-V163206(Vildagliptin Related Compound F)
The test results indicate that sample RM-V163206 demonstrates minimal variation in the main peak area during 24-hour exposure to acidic, neutral, and alkaline solutions, with relative standard deviations all below 2.0%. This confirms the sample's stability across these three pH conditions. The main peak area data for each measurement point under different pH values are as follows:

Figure 5: Summary line chart of solution stability data for sample RM-V163206 (Vildagliptin-related compound F) after 24-hour exposure to acidic, neutral, and alkaline solutions
RM-V163209(Vildagliptin Impurity I)
The test results indicate that sample RM-V163209 demonstrates minimal variation in the main peak area during 24-hour storage in acidic, neutral, and alkaline solutions, with relative standard deviations all below 2.0%. This confirms the sample's stability across these three pH conditions. The main peak area data for each measurement point under different pH values are as follows:

Figure 6: Summary line chart of solution stability data for sample RM-V163209 (Vildagliptin Impurity I) after 24-hour exposure to acidic, neutral, and alkaline solutions
3. Summary
In conclusion, this experiment demonstrates that samples RM-V163206 (Vildagliptin Related Compound F) and RM-V163209 (Vildagliptin Impurity I) exhibit excellent stability in acidic, neutral, and alkaline solutions. In contrast, RM-V163205 (Vildagliptin Related Compound E) shows good stability in acidic and neutral solutions but undergoes rapid degradation over time in alkaline solutions. Therefore, when testing sample RM-V163205 (Vildagliptin Related Compound E), customers should avoid using alkaline diluents and refrain from contact with alkaline reagents during handling, storage, and transportation. For further inquiries regarding the stability of these three samples, please contact our company.


Junte-se à nossa lista de e-mails
Inscreva-se para receber atualizações sobre novos produtos, promoções e recursos!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号