orders@qcsrm.com | +86 755-6685 3366 | 2851296953 | 13670046396
Tempo:2026-03-06

Tacrolimus (structural formula shown in Figure 1) is a potent macrolide immunosuppressant. At the molecular level, its action is mediated by the cytoplasmic protein FKBP12, which binds to it. FKBP12 facilitates the intracellular entry of the drug and forms a complex that competitively binds to calcineurin, thereby inhibiting calcineurin. Calcineurin mediates the intracellular calcium-dependent inhibitory signaling system in T cells, which blocks the transcription of a series of lymphokine genes. In vitro and in vivo experiments have demonstrated that tacrolimus is a potent immunosuppressant that inhibits the formation of cytotoxic lymphocytes, which are primarily responsible for transplant rejection.
Tacrolimus inhibits T-cell activation and TH cell-dependent B-cell proliferation, as well as the production of lymphokines such as interleukin-2 (IL-2), interleukin-3 (IL-3), and interferon-γ (IFN-γ), along with the expression of interleukin-2 receptors. Tacrolimus can suppress rejection reactions in skin, heart, kidney, and liver transplants, prolonging the survival of allografts. Its efficacy has been demonstrated in rodents, dogs, primates, and humans. Since its approval, it has been widely used for the treatment of acute and chronic rejection in liver, kidney, and bone marrow transplants.
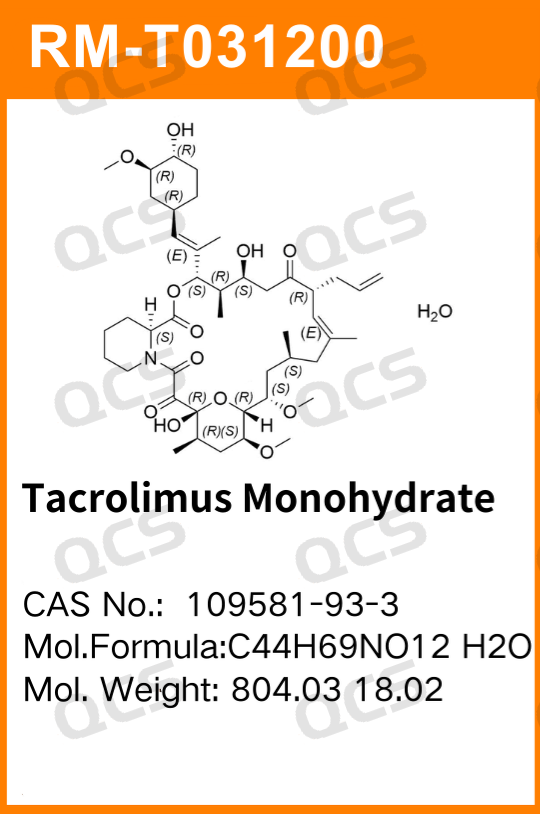
Figure 1: Structure of tacrolimus
I. R&D Background
Pharmacopoeias and registration standards of various countries have incorporated the impurity control of tacrolimus into the mandatory standard system. Mainstream pharmacopoeias such as the United States Pharmacopeia, European Pharmacopoeia, and China Pharmacopoeia have clearly specified the types, limits, and detection methods for impurities of tacrolimus, covering key quality control items such as appearance, related substances, content, and storage conditions. Different pharmacopoeias vary in impurity limits and detection methods (e.g., chromatographic column selection, mobile phase composition, detection wavelength) to accommodate regional pharmaceutical production and regulatory requirements. This article lists some chromatographic detection parameters for impurities of tacrolimus, as shown in Figure 2.
As one of our company's best-selling impurity series, tacrolimus impurities are subject to EP standard testing to ensure better quality control. This article shares empirical insights on the selection of chromatographic columns under the EP standard testing criteria.
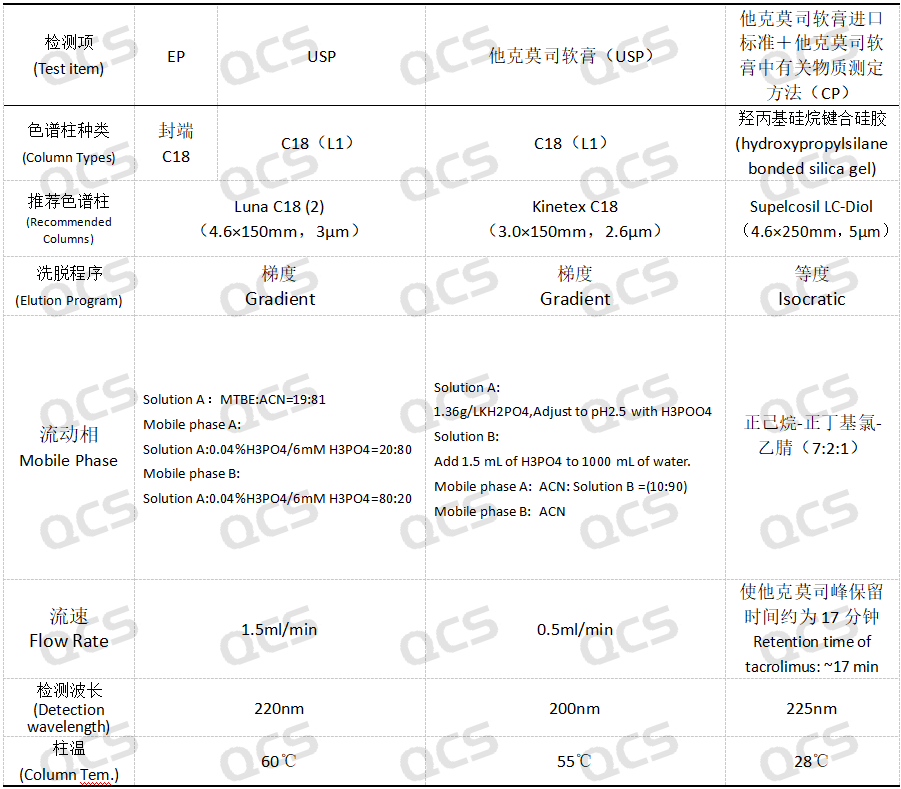
Figure 2: Summary of chromatographic detection parameters for tacrolimus under different standards (for reference substances)
2. Analysis of EP Chromatographic Conditions
After performing qualitative analysis of tacrolimus using magnetic resonance spectroscopy (NMR) and mass spectrometry, our laboratory conducted HPLC liquid-phase analysis in accordance with the substance method specified in the EP (European Pharmacopoeia) standards for tacrolimus. The EP chromatographic conditions employed a mixed solvent of methyl tert-butyl ether (MTBE) and acetonitrile as the organic phase, with aqueous phosphoric acid solution as the aqueous phase, and elution was performed under conditions of elevated column temperature and flow rate. The EP chromatographic conditions demonstrate a typical analytical approach for highly hydrophobic macrolides:
1. Using a packed C18 column and operating at 60°C can effectively improve mass transfer efficiency and reduce tailing caused by strong retention.
2. The introduction of a mixed organic system of MTBE and acetonitrile into the mobile phase significantly modulates selectivity, effectively separating structurally similar impurities. Simultaneously, the strong acidic environment of phosphoric acid suppresses the silanol group effect, optimizing peak shape and reproducibility.
3. A higher flow rate (1.5 mL/min) further reduces the analysis time.
The overall approach emphasizes separation capability and stability in complex impurity systems, reflecting the European Pharmacopoeia's (EP) technical route of "selectivity priority" in quality control for complex natural product drugs. For column selection, the EP recommends the Luna C18(2) chromatographic column, with details shown in Figures 3 and 4:

Figure 3: Tacrolimus EP recommended chromatographic column

Figure 4: Information on the recommended chromatographic column for tacrolimus EP
3. Comparison of Screening Results for Alternative Chromatographic Columns
However, if the chromatographic column recommended in the pharmacopoeia is not readily available in the laboratory or needs to be replaced for other reasons, how should a chromatographic column of a different brand or series be selected to replace the original column specified in the method?
The query results indicate that Luna C18 (2) is a conventional general-purpose C18 column, featuring an ultra-pure silica gel matrix with high binding density and uniformity. The formed "hydrophobic protective layer" exhibits strong inertness, effectively reducing the interaction between analytes and residual silanol groups, thereby improving peak shape.
Based on this, our company conducted comparative tests using five different brands of C18 chromatography columns under EP conditions, with the results shown in Figures 6-9 below.

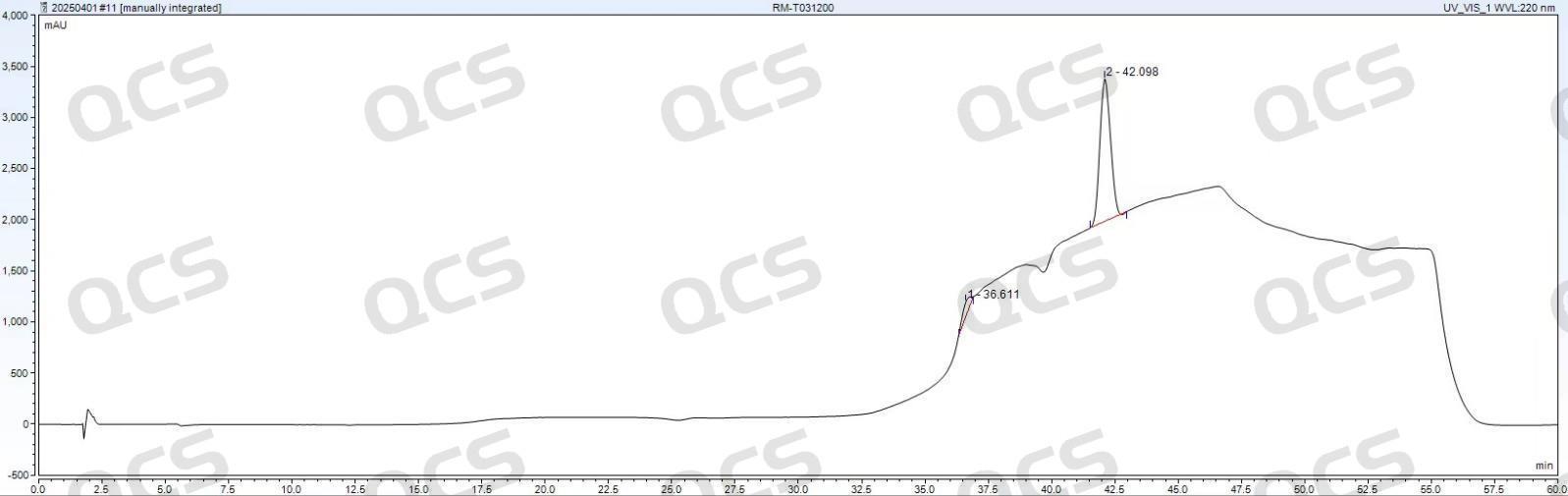
Figure 5: Zhongpu Science AQ-C18 (4.6*250mm, 5μm)
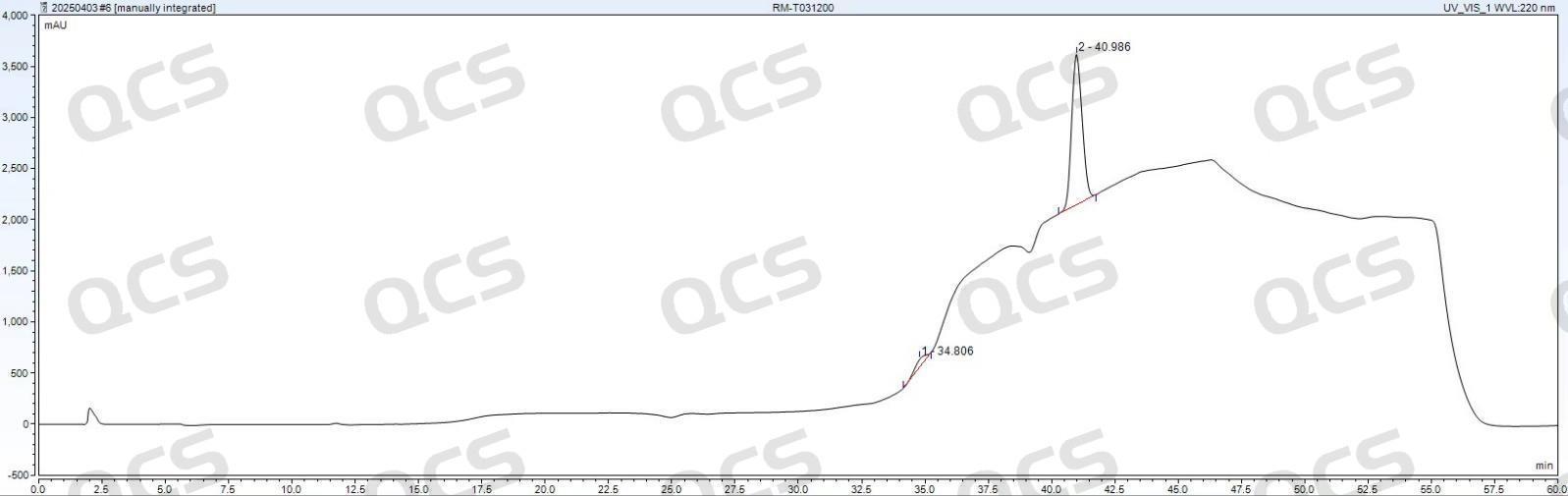
Figure 6: Zorbax SB-C18 (4.6*250mm, 5μm)
Observation of Figures 5 and 6 reveals that in the standard detection of tacrolimus EP, significant back peaks appear at the baseline position around 40 minutes when using Zhongpu Science AQ-C18 (4.6*250mm, 5μm) and Zorbax SB-C18 (4.6*250mm, 5μm). This phenomenon directly interferes with quantitative accuracy and poses two major risks:
risk ①
Baseline drift and integration error: The inverted peak causes irregular fluctuations of the baseline in the elution interval of the target peak. The integration software may mistakenly use the valley of the inverted peak as the baseline zero point, thereby underestimating the peak area of the target peak and directly leading to lower quantitative results.
risk ②
Risk of impurity peak misidentification: If the partial degradation impurities or process residues of tacrolimus happen to appear in the peak range around 40 min, the inverted negative signal may overlap with the positive signal of trace impurities, masking the presence of the true impurity peak and thereby affecting the accurate assessment of product purity.
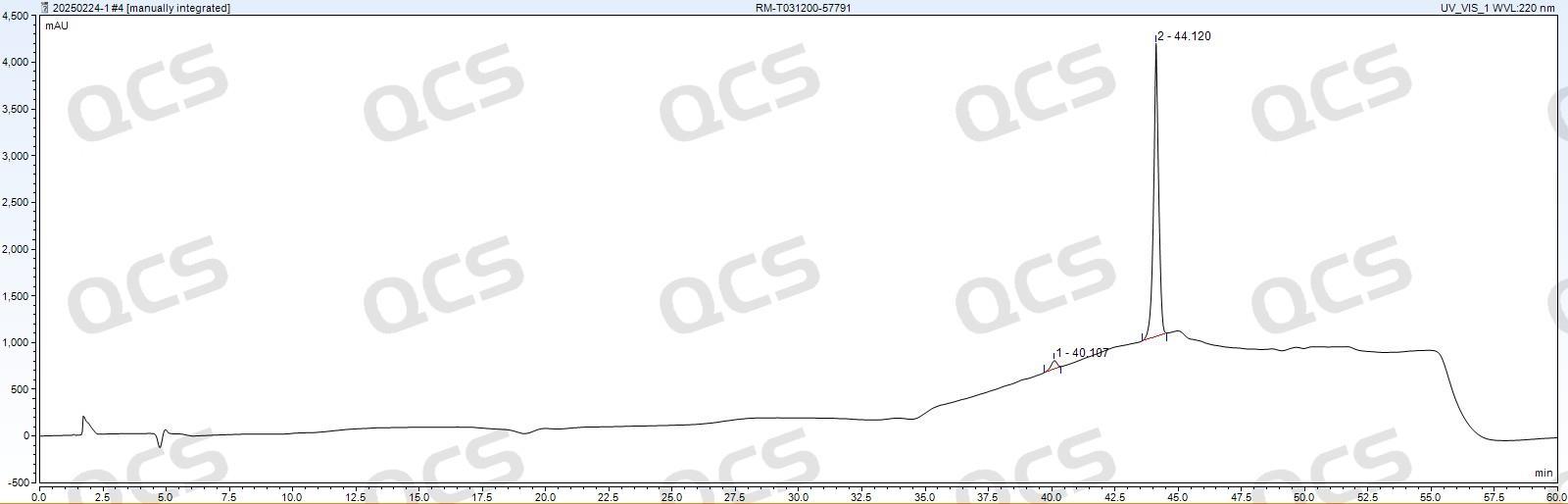
Figure 7: Luna Omega Polar C18 100A (4.6×150 mm, 5 μm)
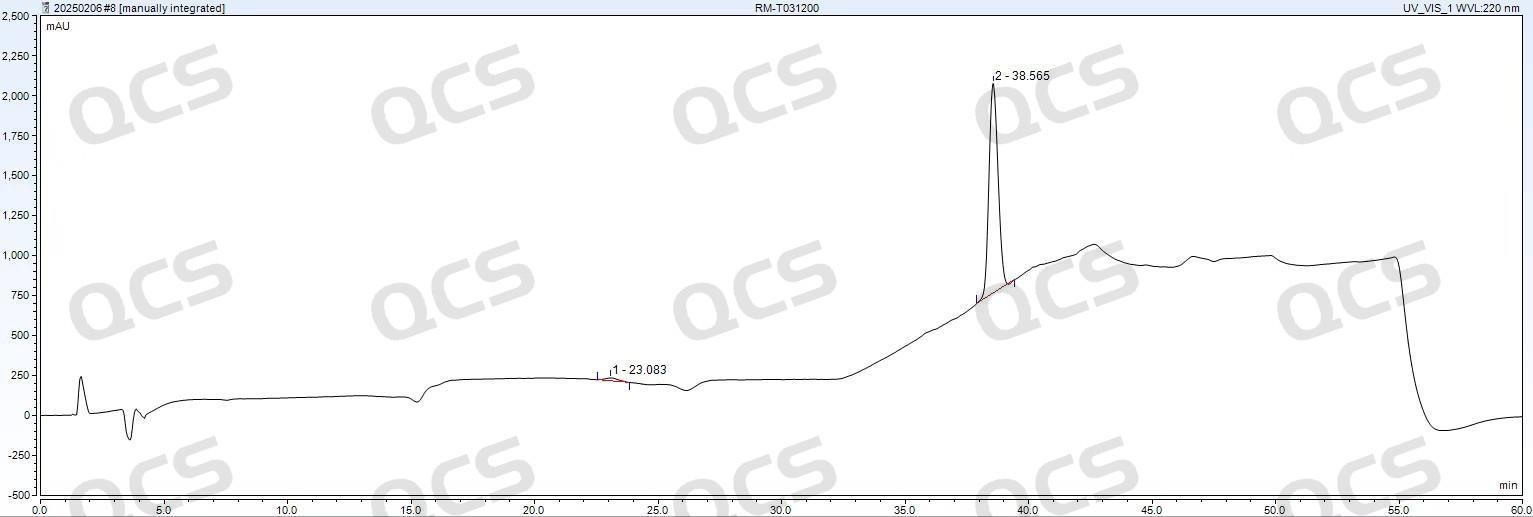
Figure 8: XBridge C18 (4.6×250 mm, 5 μm)
As shown in Figures 7 and 8, the performance of Luna Omega Polar C18 100A (4.6×150 mm, 5 μm) and Waters XBridge C18 (4.6×250 mm, 5 μm) columns is comparable in tacrolimus EP standard testing, both meeting Ep detection requirements. Given that the recommended upper temperature limit for Luna series HPLC columns is 60°C, while the Waters XBridge C18 column can operate at up to 80°C under low pH conditions, the XBridge C18 demonstrates superior high-temperature and acid resistance. This advantage reduces column replacement frequency and lowers long-term operational costs.
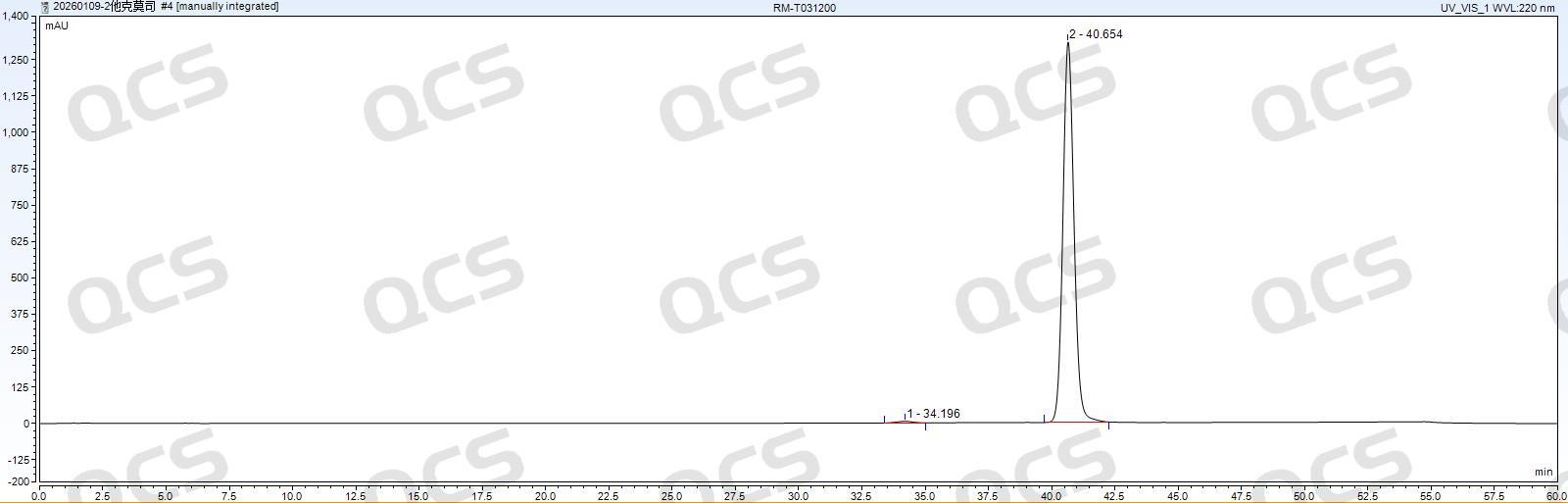
Figure 9: WondaSil C18 Superb (4.6×250 mm, 5 μm)
As shown in Figure 19, WondaSil C18 Superb (4.6×250 mm, 5 μm) demonstrated excellent performance in the standard detection of tacrolimus EP. The peak profile was sharp and symmetrical, with no tailing or fronting, exhibiting outstanding retention and separation capability for tacrolimus and high column efficiency.
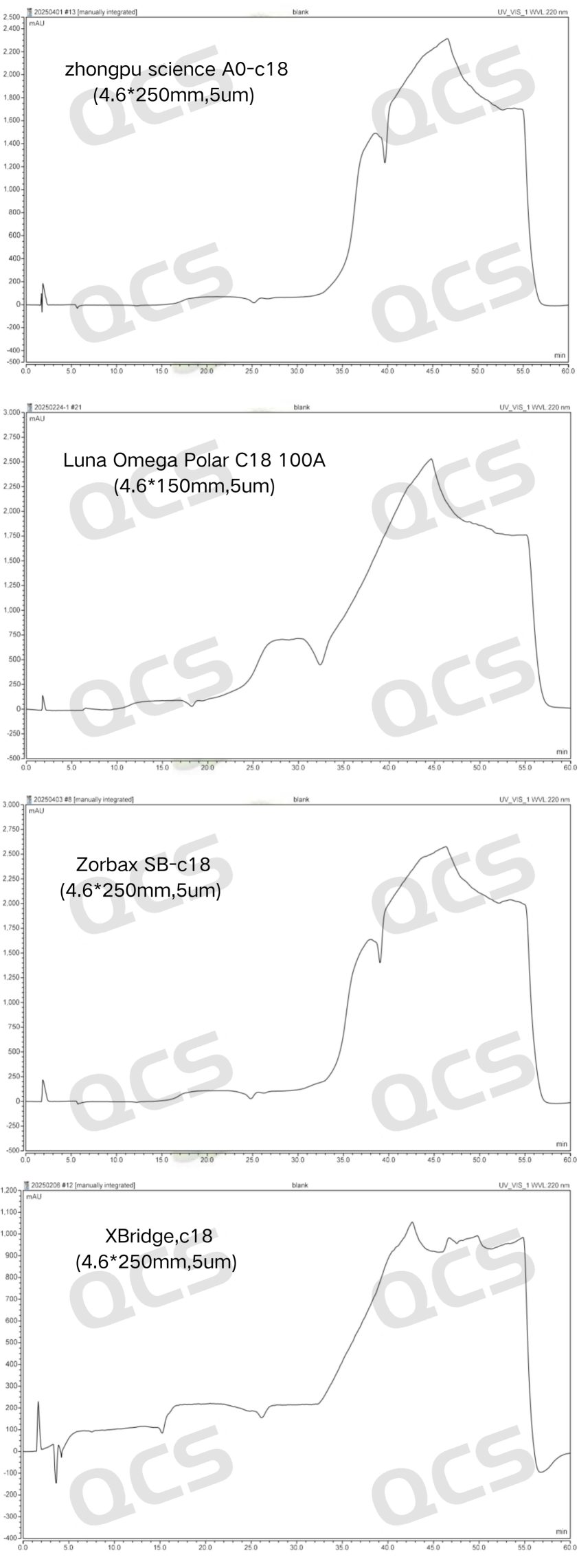
Figure 10: Blank chromatograms of four chromatographic columns
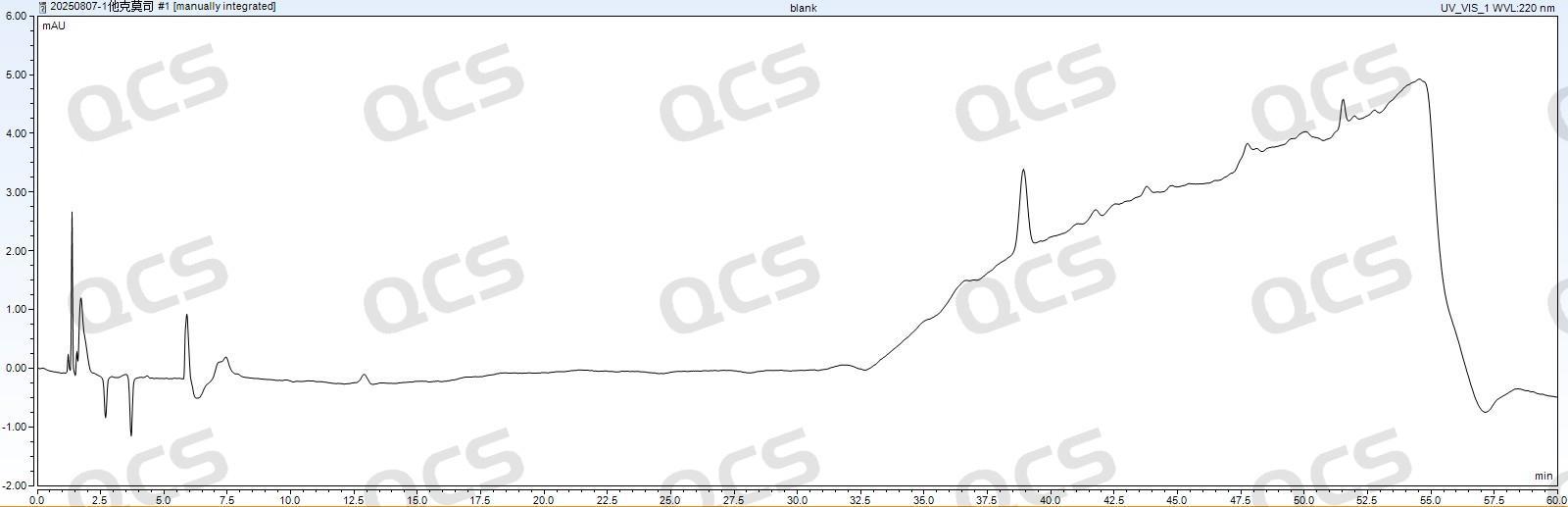
Figure 11: WondaSil C18 Superb (4.6*250mm, 5μm) blank chromatogram
As shown in Figures 10 and 11, the blank baseline response of four chromatographic columns—Zhongpu Science AQ-C18 (4.6*250mm, 5μm), Zorbax SB-C18 (4.6*250mm, 5μm), Luna Omega Polar C18 100A (4.6*150mm, 5μm), and XBridge C18 (4.6*250mm, 5μm)—ranged from 1000mAU to 2500mAU. In contrast, the blank baseline response of WondaSil C18 Superb (4.6*250mm, 5μm) was controllable below 10mAU, with a stable baseline free from significant drift or noise. No inverted peaks or interfering signals were observed before or after the target peak, and the system background interference was extremely low, providing a reliable foundation for accurate integration. Additionally, the detection personnel could more intuitively observe the test results.
4. Summary
Tacrolimus is a macrolide immunosuppressant that lacks a strongly conjugated chromophore system in its molecular structure. Its primary ultraviolet absorption originates from carbonyl (C=O), hydroxyl (-OH), and isolated double bonds within the molecule, with all these groups exhibiting absorption within the terminal absorption region. Consequently, the maximum absorption wavelength of tacrolimus typically ranges between 210 nm and 220 nm.
For the ultraviolet detection wavelength range of 190 nm to 220 nm, low-wavelength dedicated columns are typically employed. When analyzing samples at low wavelengths, chromatography column manufacturers often introduce column series labeled with "low backgrounds" or similar specifications. The key features include high-purity silica gel matrix fillers with minimal bonded phase residues, which exhibit no additional absorption at low UV wavelengths, thereby preventing baseline drift or interference.
This study compared five chromatographic columns: Zhongpu Science AQ-C18 (4.6*250mm, 5μm), Zorbax SB-C18 (4.6*250mm, 5μm), Luna Omega Polar C18 100A (4.6*150mm, 5μm), XBridge C18 (4.6*250mm, 5μm), and WondaSil C18 Superb (4.6*250mm, 5μm). The experimental results demonstrated that WondaSil C18 Superb (4.6*250mm, 5μm) exhibited superior performance in tacrolimus detection. Therefore, if column selection is required for tacrolimus detection, WondaSil C18 Superb (4.6*250mm, 5μm) is recommended as the preferred option.



Tacrolimus (structural formula shown in Figure 1) is a potent macrolide immunosuppressant. At the molecular level, its action is mediated by the cytoplasmic protein FKBP12, which binds to it. FKBP12 facilitates the intracellular entry of the drug and forms a complex that competitively binds to calcineurin, thereby inhibiting calcineurin. Calcineurin mediates the intracellular calcium-dependent inhibitory signaling system in T cells, which blocks the transcription of a series of lymphokine genes. In vitro and in vivo experiments have demonstrated that tacrolimus is a potent immunosuppressant that inhibits the formation of cytotoxic lymphocytes, which are primarily responsible for transplant rejection.
Tacrolimus inhibits T-cell activation and TH cell-dependent B-cell proliferation, as well as the production of lymphokines such as interleukin-2 (IL-2), interleukin-3 (IL-3), and interferon-γ (IFN-γ), along with the expression of interleukin-2 receptors. Tacrolimus can suppress rejection reactions in skin, heart, kidney, and liver transplants, prolonging the survival of allografts. Its efficacy has been demonstrated in rodents, dogs, primates, and humans. Since its approval, it has been widely used for the treatment of acute and chronic rejection in liver, kidney, and bone marrow transplants.

Figure 1: Structure of tacrolimus
I. R&D Background
Pharmacopoeias and registration standards of various countries have incorporated the impurity control of tacrolimus into the mandatory standard system. Mainstream pharmacopoeias such as the United States Pharmacopeia, European Pharmacopoeia, and China Pharmacopoeia have clearly specified the types, limits, and detection methods for impurities of tacrolimus, covering key quality control items such as appearance, related substances, content, and storage conditions. Different pharmacopoeias vary in impurity limits and detection methods (e.g., chromatographic column selection, mobile phase composition, detection wavelength) to accommodate regional pharmaceutical production and regulatory requirements. This article lists some chromatographic detection parameters for impurities of tacrolimus, as shown in Figure 2.
As one of our company's best-selling impurity series, tacrolimus impurities are subject to EP standard testing to ensure better quality control. This article shares empirical insights on the selection of chromatographic columns under the EP standard testing criteria.

Figure 2: Summary of chromatographic detection parameters for tacrolimus under different standards (for reference substances)
2. Analysis of EP Chromatographic Conditions
After performing qualitative analysis of tacrolimus using magnetic resonance spectroscopy (NMR) and mass spectrometry, our laboratory conducted HPLC liquid-phase analysis in accordance with the substance method specified in the EP (European Pharmacopoeia) standards for tacrolimus. The EP chromatographic conditions employed a mixed solvent of methyl tert-butyl ether (MTBE) and acetonitrile as the organic phase, with aqueous phosphoric acid solution as the aqueous phase, and elution was performed under conditions of elevated column temperature and flow rate. The EP chromatographic conditions demonstrate a typical analytical approach for highly hydrophobic macrolides:
1. Using a packed C18 column and operating at 60°C can effectively improve mass transfer efficiency and reduce tailing caused by strong retention.
2. The introduction of a mixed organic system of MTBE and acetonitrile into the mobile phase significantly modulates selectivity, effectively separating structurally similar impurities. Simultaneously, the strong acidic environment of phosphoric acid suppresses the silanol group effect, optimizing peak shape and reproducibility.
3. A higher flow rate (1.5 mL/min) further reduces the analysis time.
The overall approach emphasizes separation capability and stability in complex impurity systems, reflecting the European Pharmacopoeia's (EP) technical route of "selectivity priority" in quality control for complex natural product drugs. For column selection, the EP recommends the Luna C18(2) chromatographic column, with details shown in Figures 3 and 4:

Figure 3: Tacrolimus EP recommended chromatographic column

Figure 4: Information on the recommended chromatographic column for tacrolimus EP
3. Comparison of Screening Results for Alternative Chromatographic Columns
However, if the chromatographic column recommended in the pharmacopoeia is not readily available in the laboratory or needs to be replaced for other reasons, how should a chromatographic column of a different brand or series be selected to replace the original column specified in the method?
The query results indicate that Luna C18 (2) is a conventional general-purpose C18 column, featuring an ultra-pure silica gel matrix with high binding density and uniformity. The formed "hydrophobic protective layer" exhibits strong inertness, effectively reducing the interaction between analytes and residual silanol groups, thereby improving peak shape.
Based on this, our company conducted comparative tests using five different brands of C18 chromatography columns under EP conditions, with the results shown in Figures 6-9 below.


Figure 5: Zhongpu Science AQ-C18 (4.6*250mm, 5μm)

Figure 6: Zorbax SB-C18 (4.6*250mm, 5μm)
Observation of Figures 5 and 6 reveals that in the standard detection of tacrolimus EP, significant back peaks appear at the baseline position around 40 minutes when using Zhongpu Science AQ-C18 (4.6*250mm, 5μm) and Zorbax SB-C18 (4.6*250mm, 5μm). This phenomenon directly interferes with quantitative accuracy and poses two major risks:
risk ①
Baseline drift and integration error: The inverted peak causes irregular fluctuations of the baseline in the elution interval of the target peak. The integration software may mistakenly use the valley of the inverted peak as the baseline zero point, thereby underestimating the peak area of the target peak and directly leading to lower quantitative results.
risk ②
Risk of impurity peak misidentification: If the partial degradation impurities or process residues of tacrolimus happen to appear in the peak range around 40 min, the inverted negative signal may overlap with the positive signal of trace impurities, masking the presence of the true impurity peak and thereby affecting the accurate assessment of product purity.

Figure 7: Luna Omega Polar C18 100A (4.6×150 mm, 5 μm)

Figure 8: XBridge C18 (4.6×250 mm, 5 μm)
As shown in Figures 7 and 8, the performance of Luna Omega Polar C18 100A (4.6×150 mm, 5 μm) and Waters XBridge C18 (4.6×250 mm, 5 μm) columns is comparable in tacrolimus EP standard testing, both meeting Ep detection requirements. Given that the recommended upper temperature limit for Luna series HPLC columns is 60°C, while the Waters XBridge C18 column can operate at up to 80°C under low pH conditions, the XBridge C18 demonstrates superior high-temperature and acid resistance. This advantage reduces column replacement frequency and lowers long-term operational costs.

Figure 9: WondaSil C18 Superb (4.6×250 mm, 5 μm)
As shown in Figure 19, WondaSil C18 Superb (4.6×250 mm, 5 μm) demonstrated excellent performance in the standard detection of tacrolimus EP. The peak profile was sharp and symmetrical, with no tailing or fronting, exhibiting outstanding retention and separation capability for tacrolimus and high column efficiency.

Figure 10: Blank chromatograms of four chromatographic columns

Figure 11: WondaSil C18 Superb (4.6*250mm, 5μm) blank chromatogram
As shown in Figures 10 and 11, the blank baseline response of four chromatographic columns—Zhongpu Science AQ-C18 (4.6*250mm, 5μm), Zorbax SB-C18 (4.6*250mm, 5μm), Luna Omega Polar C18 100A (4.6*150mm, 5μm), and XBridge C18 (4.6*250mm, 5μm)—ranged from 1000mAU to 2500mAU. In contrast, the blank baseline response of WondaSil C18 Superb (4.6*250mm, 5μm) was controllable below 10mAU, with a stable baseline free from significant drift or noise. No inverted peaks or interfering signals were observed before or after the target peak, and the system background interference was extremely low, providing a reliable foundation for accurate integration. Additionally, the detection personnel could more intuitively observe the test results.
4. Summary
Tacrolimus is a macrolide immunosuppressant that lacks a strongly conjugated chromophore system in its molecular structure. Its primary ultraviolet absorption originates from carbonyl (C=O), hydroxyl (-OH), and isolated double bonds within the molecule, with all these groups exhibiting absorption within the terminal absorption region. Consequently, the maximum absorption wavelength of tacrolimus typically ranges between 210 nm and 220 nm.
For the ultraviolet detection wavelength range of 190 nm to 220 nm, low-wavelength dedicated columns are typically employed. When analyzing samples at low wavelengths, chromatography column manufacturers often introduce column series labeled with "low backgrounds" or similar specifications. The key features include high-purity silica gel matrix fillers with minimal bonded phase residues, which exhibit no additional absorption at low UV wavelengths, thereby preventing baseline drift or interference.
This study compared five chromatographic columns: Zhongpu Science AQ-C18 (4.6*250mm, 5μm), Zorbax SB-C18 (4.6*250mm, 5μm), Luna Omega Polar C18 100A (4.6*150mm, 5μm), XBridge C18 (4.6*250mm, 5μm), and WondaSil C18 Superb (4.6*250mm, 5μm). The experimental results demonstrated that WondaSil C18 Superb (4.6*250mm, 5μm) exhibited superior performance in tacrolimus detection. Therefore, if column selection is required for tacrolimus detection, WondaSil C18 Superb (4.6*250mm, 5μm) is recommended as the preferred option.


Junte-se à nossa lista de e-mails
Inscreva-se para receber atualizações sobre novos produtos, promoções e recursos!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号