orders@qcsrm.com | +86 755-6685 3366 | 2851296953 | 13670046396
Tempo:2026-03-12


Product/Introduction:
Beritinib (brand name: Wanbirui) is a highly selective c-MET kinase inhibitor administered orally that crosses the blood-brain barrier.
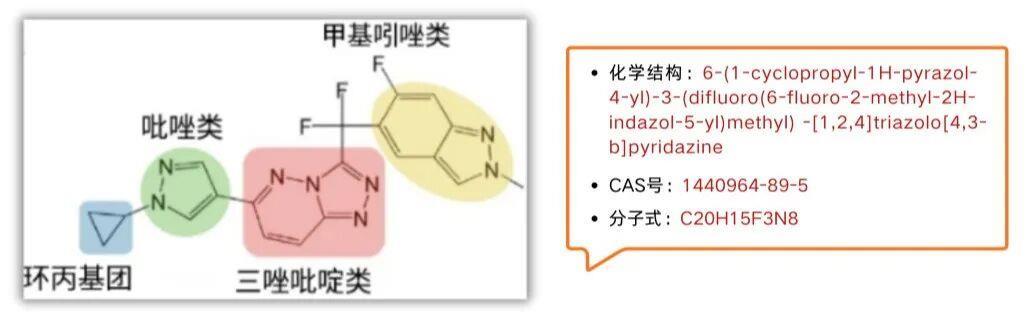
01 Mechanism of Action of Beritinib
Unlike the lymphocyte "gate" regulation mechanism of itraconazole, the action mechanism of beratinib resembles a precise "molecular key." It selectively binds with high affinity to the ATP-binding pocket of the c-MET kinase, thereby blocking abnormal phosphorylation induced by METex14 mutations or PTPRZ1-MET fusions. By inhibiting c-MET phosphorylation, it disrupts downstream signaling pathways such as RAS-RAF-MEK-ERK and PI3K-AKT, leading to suppression of tumor cell proliferation (G0/G1 phase arrest), induction of tumor cell apoptosis, and inhibition of tumor angiogenesis, invasion, and metastasis.
Especially in the treatment of gliomas, its exceptional ability to penetrate the blood-brain barrier enables drugs to reach intracranial lesions and suppress the driving gene signals at their source.
02 Listing Overview
With twice-daily oral administration, high intracranial exposure, and excellent safety profile, berutinib has achieved pivotal progress in the China market:

This is the first fully approved small-molecule targeted drug in China for MET-targeted therapy of gliomas.

03 Contaminant Study: Control System Based on ICH Guidelines
With the increasing focus on the domestic development and generic drug deployment of berutinib, impurity profile analysis and quality control have become pivotal for IND/NDA submissions and consistency evaluations. Given that berutinib lacks chiral centers, impurity control primarily targets genotoxic impurities and process-related impurities.
Process-related impurities (genotoxicity impurity risk)
Potential nitrosamine impurities (NDSRIs)
Forced degradation of impurities (stability indicator)
Residual solvent and elemental impurities
Impurity studies require reference standards with confirmed structures, which encompass process impurities, intermediates, and specific degradation impurities generated by forced degradation. Currently, the QCS can provide multiple bozitinib-related impurities (as shown below):
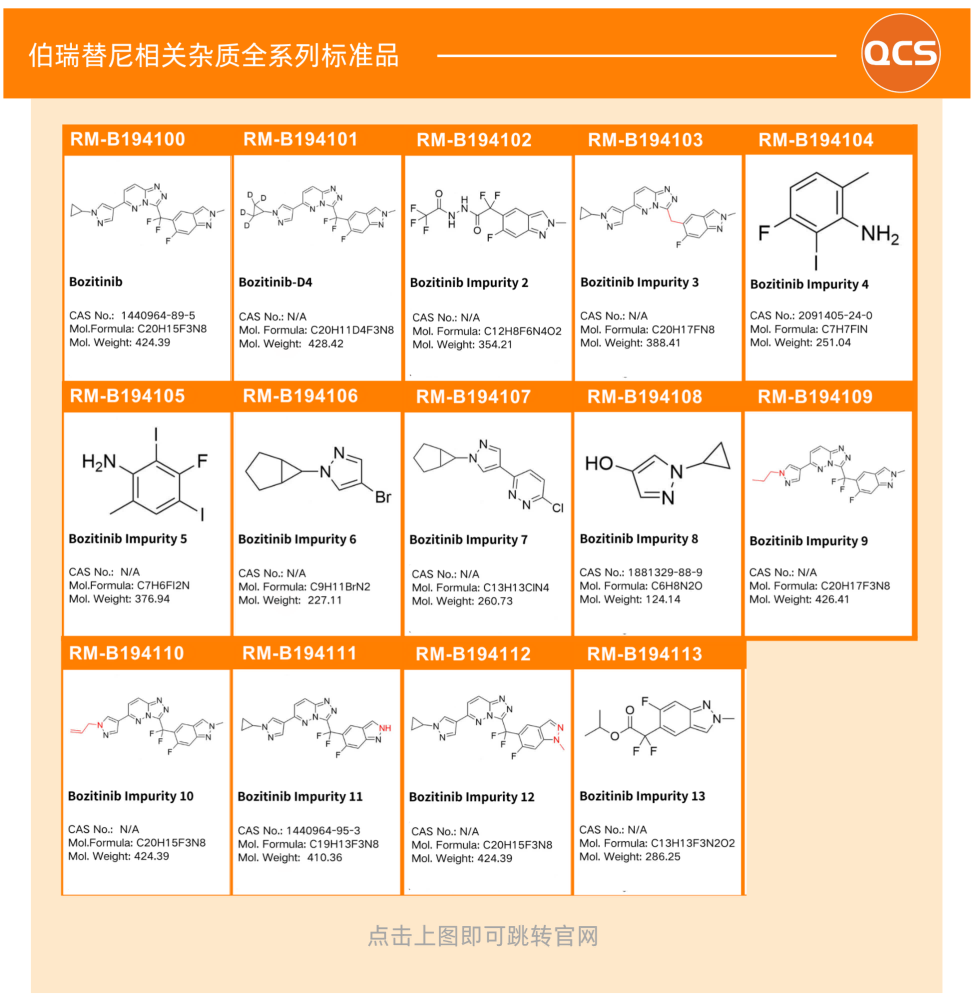
All impurity products are accompanied by complete Quality Certificate of Analysis (COA), containing structural confirmation data such as NMR and MS results, as well as purity determination results. The QCS has obtained ANAB ISO 17034 accreditation, with its standard preparation and calibration processes meeting the international requirements for reference substance manufacturer certification. This ensures that the data provided can be internationally recognized for regulatory submissions to agencies such as the NMPA, EMA, and FDA.





Product/Introduction:
Beritinib (brand name: Wanbirui) is a highly selective c-MET kinase inhibitor administered orally that crosses the blood-brain barrier.

01 Mechanism of Action of Beritinib
Unlike the lymphocyte "gate" regulation mechanism of itraconazole, the action mechanism of beratinib resembles a precise "molecular key." It selectively binds with high affinity to the ATP-binding pocket of the c-MET kinase, thereby blocking abnormal phosphorylation induced by METex14 mutations or PTPRZ1-MET fusions. By inhibiting c-MET phosphorylation, it disrupts downstream signaling pathways such as RAS-RAF-MEK-ERK and PI3K-AKT, leading to suppression of tumor cell proliferation (G0/G1 phase arrest), induction of tumor cell apoptosis, and inhibition of tumor angiogenesis, invasion, and metastasis.
Especially in the treatment of gliomas, its exceptional ability to penetrate the blood-brain barrier enables drugs to reach intracranial lesions and suppress the driving gene signals at their source.
02 Listing Overview
With twice-daily oral administration, high intracranial exposure, and excellent safety profile, berutinib has achieved pivotal progress in the China market:

This is the first fully approved small-molecule targeted drug in China for MET-targeted therapy of gliomas.

03 Contaminant Study: Control System Based on ICH Guidelines
With the increasing focus on the domestic development and generic drug deployment of berutinib, impurity profile analysis and quality control have become pivotal for IND/NDA submissions and consistency evaluations. Given that berutinib lacks chiral centers, impurity control primarily targets genotoxic impurities and process-related impurities.
Process-related impurities (genotoxicity impurity risk)
Potential nitrosamine impurities (NDSRIs)
Forced degradation of impurities (stability indicator)
Residual solvent and elemental impurities
Impurity studies require reference standards with confirmed structures, which encompass process impurities, intermediates, and specific degradation impurities generated by forced degradation. Currently, the QCS can provide multiple bozitinib-related impurities (as shown below):

All impurity products are accompanied by complete Quality Certificate of Analysis (COA), containing structural confirmation data such as NMR and MS results, as well as purity determination results. The QCS has obtained ANAB ISO 17034 accreditation, with its standard preparation and calibration processes meeting the international requirements for reference substance manufacturer certification. This ensures that the data provided can be internationally recognized for regulatory submissions to agencies such as the NMPA, EMA, and FDA.



Junte-se à nossa lista de e-mails
Inscreva-se para receber atualizações sobre novos produtos, promoções e recursos!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号