orders@qcsrm.com | +86 755-6685 3366 | 2851296953 | 13670046396
Tempo:2026-03-25

Product/Introduction: From "Immunological Dysregulation" to Targeted Intervention for Chronic Spontaneous Urticaria
Chronic spontaneous urticaria (CSU) is a complex inflammatory skin disorder characterized by recurrent wheals and angioedema without identifiable triggers, often accompanied by sleep disturbances, emotional distress, and decreased quality of life. Traditional treatment primarily involves second-generation H1 antihistamines, yet more than half of patients exhibit suboptimal symptom control. Subsequent second-line therapy with omalizumab (an anti-IgE monoclonal antibody) is effective; however, its injectable formulation poses challenges in terms of adherence and cost, and some patients still demonstrate inadequate response.
It is against this backdrop that remibrutinib, a next-generation orally administered, highly selective covalent Bruton tyrosine kinase (BTK) inhibitor, has represented a breakthrough advancement in the treatment of CSU. Developed by Novartis, it is marketed under the brand name Rhapsido®.
01 Mechanism of Action of Ributinib
Unlike the "symptom suppression" mechanism of traditional antihistamines, the therapeutic rationale of ributinib resembles a "molecular switch" that precisely blocks inflammatory signals. The core pathological mechanism of chronic spontaneous asthma involves the abnormal activation of mast cells and basophils through high-affinity IgE receptors (FcεRI), which subsequently activates the downstream BTK signaling pathway, leading to the release of histamine and inflammatory cytokines.
Ributinib, as a highly selective covalent BTK inhibitor, irreversibly binds to and inhibits BTK, thereby blocking mast cell degranulation and the release of inflammatory mediators upstream, thus controlling the occurrence of wheals and pruritus.

02 Listing Overview
September 30,2025: Approved for marketing by the U.S. FDA (brand name Rhapsido®) for the treatment of chronic spontaneous urticaria in adults, making it the first oral therapeutic agent for chronic spontaneous urticaria (CSU), indicated for adults with persistent symptoms despite H1 antihistamine therapy.
February 2026: Novartis announced that its Phase III RemIND study for chronic inducible urticaria (CIndU) had met the primary endpoint and submitted a Supplementary New Drug Application (sNDA) to the FDA, positioning it as the first targeted therapy in the CIndU field.
China market: Multiple Phase III clinical trials have been conducted in China (e.g., for CSU and CIndU), while concurrent research and development are underway for immune-mediated diseases such as hidradenitis suppurativa (HS).
03 Contaminant Study: Control System Based on ICH Guidelines
Based on the molecular structural characteristics of remibrutinib (covalent acrylamide warhead, multiple amide bonds, and bifluoro substitution), its impurity control system should focus on the following four core dimensions:
1. Process-related impurities (genotoxicity impurity risk)
2. Potential nitrosamine impurities (NDSRIs)
3. Compulsory degradation impurities (stability indicator)
4. Residual solvent and elemental impurities
Impurity studies require reference standards with confirmed structures, covering process impurities, key intermediates, and specific degradation impurities generated by mandatory degradation. Currently, the QCS can provide multiple remibrutinib-related impurities (as shown below):
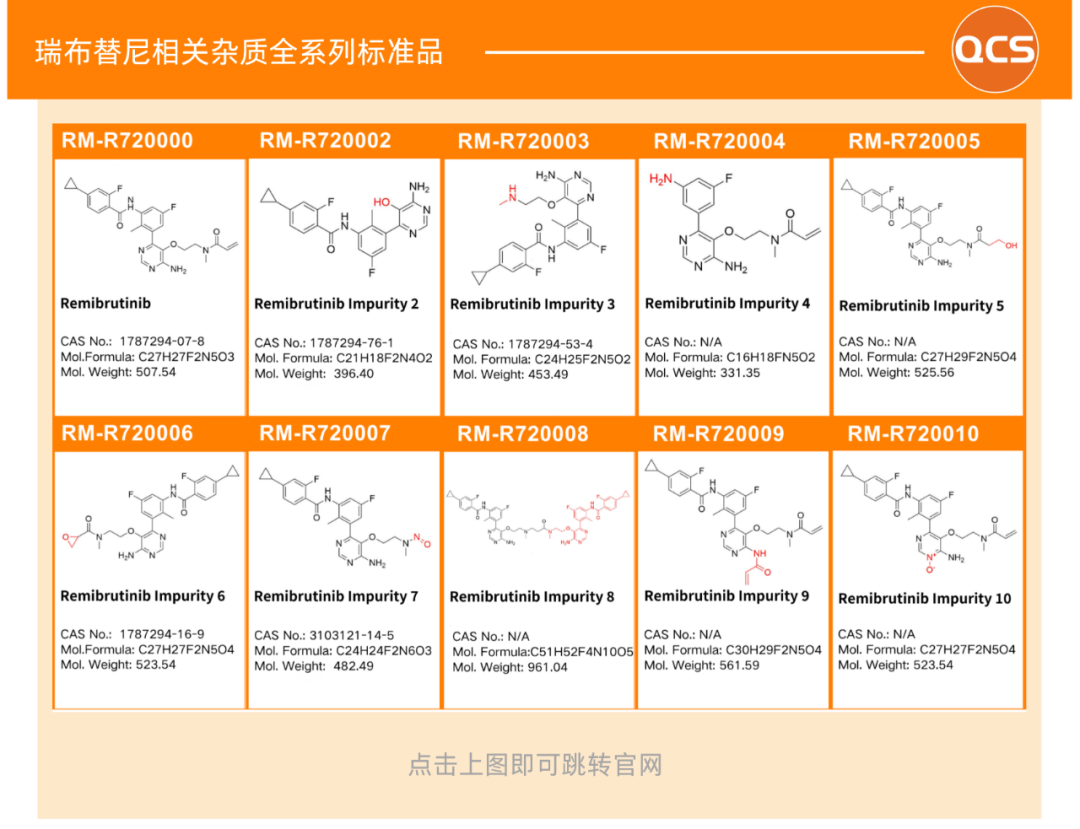
All impurity products are accompanied by complete Quality Certificate of Analysis (COA), containing structural confirmation data such as NMR and MS results, as well as purity determination results. The QCS has obtained ANAB ISO 17034 accreditation, with its standard preparation and calibration processes meeting the international requirements for reference substance manufacturer certification. This ensures that the data provided can be internationally recognized for regulatory submissions to agencies such as the NMPA, EMA, and FDA.




Product/Introduction: From "Immunological Dysregulation" to Targeted Intervention for Chronic Spontaneous Urticaria
Chronic spontaneous urticaria (CSU) is a complex inflammatory skin disorder characterized by recurrent wheals and angioedema without identifiable triggers, often accompanied by sleep disturbances, emotional distress, and decreased quality of life. Traditional treatment primarily involves second-generation H1 antihistamines, yet more than half of patients exhibit suboptimal symptom control. Subsequent second-line therapy with omalizumab (an anti-IgE monoclonal antibody) is effective; however, its injectable formulation poses challenges in terms of adherence and cost, and some patients still demonstrate inadequate response.
It is against this backdrop that remibrutinib, a next-generation orally administered, highly selective covalent Bruton tyrosine kinase (BTK) inhibitor, has represented a breakthrough advancement in the treatment of CSU. Developed by Novartis, it is marketed under the brand name Rhapsido®.

01 Mechanism of Action of Ributinib
Unlike the "symptom suppression" mechanism of traditional antihistamines, the therapeutic rationale of ributinib resembles a "molecular switch" that precisely blocks inflammatory signals. The core pathological mechanism of chronic spontaneous asthma involves the abnormal activation of mast cells and basophils through high-affinity IgE receptors (FcεRI), which subsequently activates the downstream BTK signaling pathway, leading to the release of histamine and inflammatory cytokines.
Ributinib, as a highly selective covalent BTK inhibitor, irreversibly binds to and inhibits BTK, thereby blocking mast cell degranulation and the release of inflammatory mediators upstream, thus controlling the occurrence of wheals and pruritus.

02 Listing Overview
September 30,2025: Approved for marketing by the U.S. FDA (brand name Rhapsido®) for the treatment of chronic spontaneous urticaria in adults, making it the first oral therapeutic agent for chronic spontaneous urticaria (CSU), indicated for adults with persistent symptoms despite H1 antihistamine therapy.
February 2026: Novartis announced that its Phase III RemIND study for chronic inducible urticaria (CIndU) had met the primary endpoint and submitted a Supplementary New Drug Application (sNDA) to the FDA, positioning it as the first targeted therapy in the CIndU field.
China market: Multiple Phase III clinical trials have been conducted in China (e.g., for CSU and CIndU), while concurrent research and development are underway for immune-mediated diseases such as hidradenitis suppurativa (HS).
03 Contaminant Study: Control System Based on ICH Guidelines
Based on the molecular structural characteristics of remibrutinib (covalent acrylamide warhead, multiple amide bonds, and bifluoro substitution), its impurity control system should focus on the following four core dimensions:
1. Process-related impurities (genotoxicity impurity risk)
2. Potential nitrosamine impurities (NDSRIs)
3. Compulsory degradation impurities (stability indicator)
4. Residual solvent and elemental impurities
Impurity studies require reference standards with confirmed structures, covering process impurities, key intermediates, and specific degradation impurities generated by mandatory degradation. Currently, the QCS can provide multiple remibrutinib-related impurities (as shown below):

All impurity products are accompanied by complete Quality Certificate of Analysis (COA), containing structural confirmation data such as NMR and MS results, as well as purity determination results. The QCS has obtained ANAB ISO 17034 accreditation, with its standard preparation and calibration processes meeting the international requirements for reference substance manufacturer certification. This ensures that the data provided can be internationally recognized for regulatory submissions to agencies such as the NMPA, EMA, and FDA.



Junte-se à nossa lista de e-mails
Inscreva-se para receber atualizações sobre novos produtos, promoções e recursos!
Join Our Email List
Subscribe to receive updates on new
products, promotions and resources!
| ISO 17034:2016 |
| ISO 9001:2015 |

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.

*Todos os produtos desta empresa são destinados apenas para pesquisa científica.
Copyright © 2021-2024 QCSRM All rights reserved. 粤ICP备2023004355号
Copyright © 2021-2024 QCSRM All rights reserved.
粤ICP备2023004355号